Researchers at Tampere University in collaboration with The University of Hong Kong have developed a light-activated drug delivery platform that enables controlled drug release using deep-penetrating red/near-infrared light addressing a major challenge in photopharmacology.
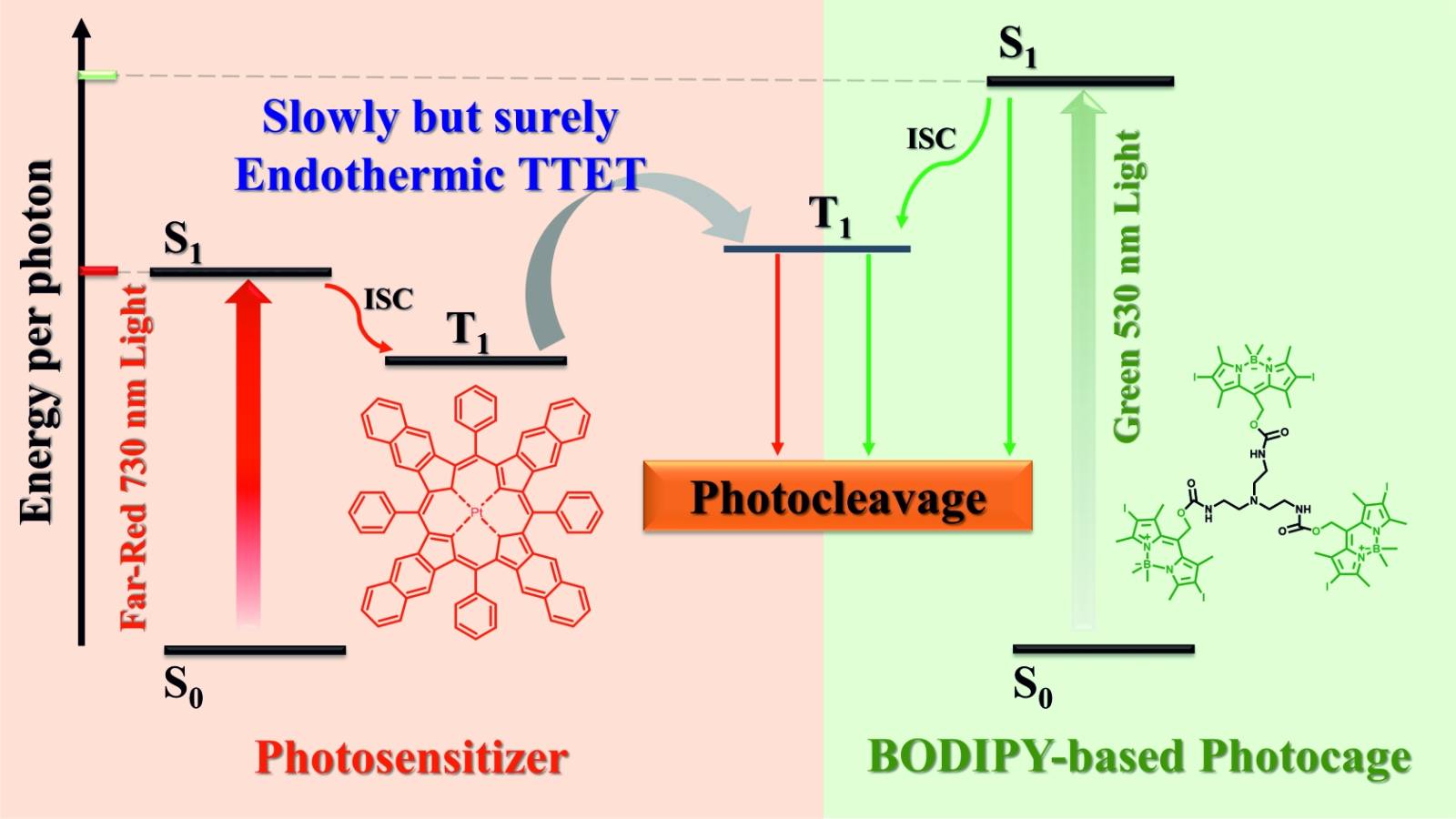
In their recent publication in Advanced Optical Materials, the team introduces a self-assembled nanovehicle built from BODIPY-based photocleavable trigonal molecules. The system incorporates a Pt(II) meso-tetraphenyltetranaphthoporphyrin photosensitizer together with a fluorescent release marker (Nile Red) in its hydrophobic core.
What makes this approach particularly powerful is its energy-transfer mechanism. Instead of directly exciting the photocleavable group with green light (530 nm), the system uses endothermic triplet-triplet energy transfer to trigger photocleavage with 730 nm light. This wavelength lies within the phototherapeutic window, offering deeper tissue penetration and safer activation without chemically modifying the photocage structure.
In cell studies using PC3 human prostate cancer cells, the nanovehicles showed rapid intracellular uptake and lysosomal accumulation within two hours. Upon 730 nm irradiation, efficient payload release was observed. Notably, the system demonstrated even higher release efficiency under low oxygen conditions, making it especially promising for treating aggressive hypoxic solid tumors.
This work presents a significant step toward safe, precise, and clinically relevant light-controlled drug delivery systems combining smart molecular design with biologically compatible activation wavelengths.
Read the full article here:
https://advanced.onlinelibrary.wiley.com/doi/10.1002/adom.202400310