Researchers from Tampere University and University of Helsinki in collaboration with Aalto University and King’s College London have provided new insights into the interactions between doxorubicin (DOX) and DNA origami nanostructures (DONs), helping to improve the design and safety of DNA-based nanocarriers for cancer therapy. The work was published in ACS Applied Nano Materials journal.
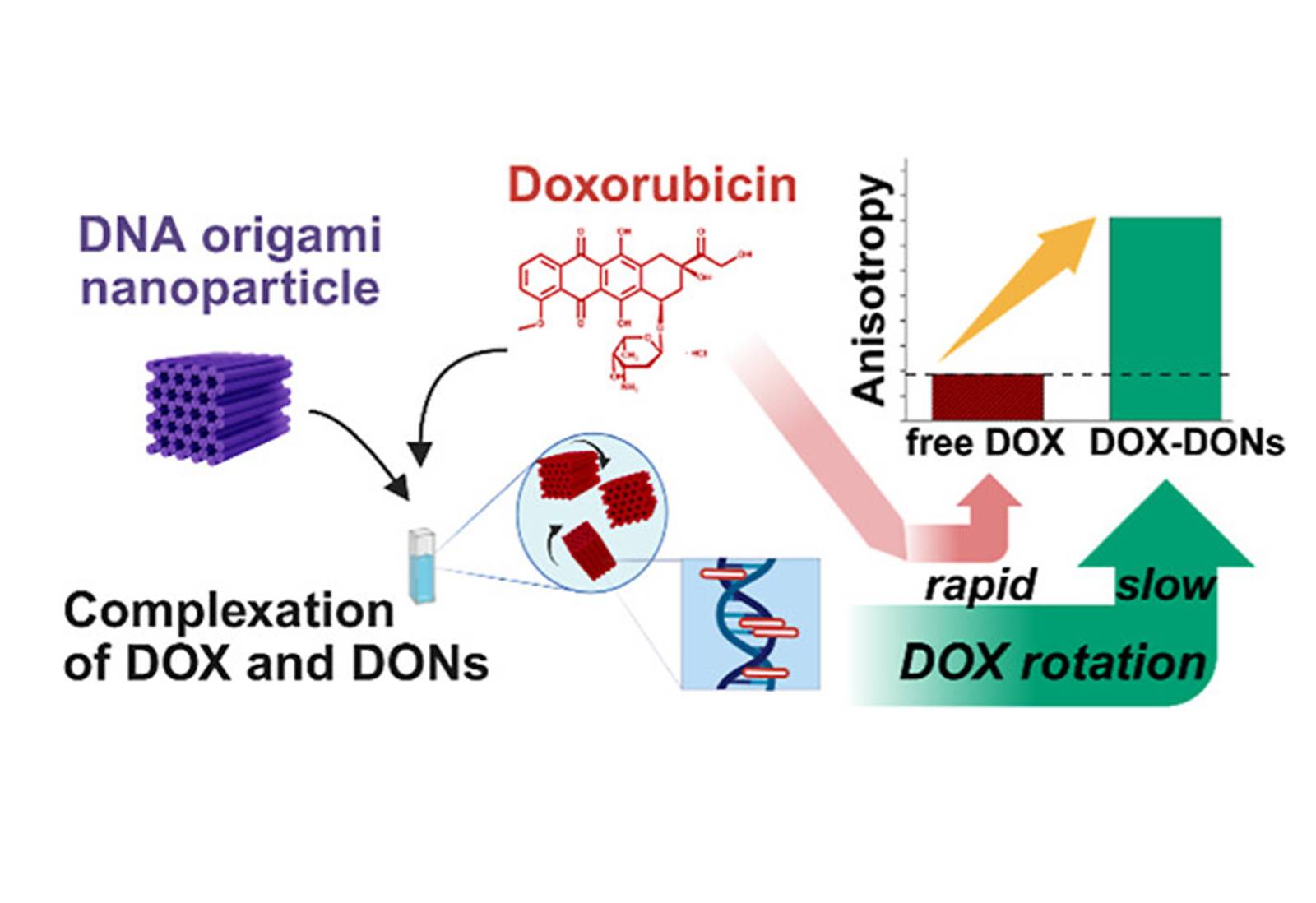
Doxorubicin is widely used in chemotherapy and has a strong natural affinity for DNA. This makes DOX-loaded DNA origami nanostructures (DOX-DONs) promising candidates for targeted drug delivery. However, the interaction between DOX and DNA origami is often assumed rather than carefully characterized and the influence of drug loading conditions has remained insufficiently understood.
In this study, the team exploited the inherent fluorescence of doxorubicin to investigate DOX-DON interactions using steady-state and time-resolved fluorescence anisotropy spectroscopy. This non-destructive approach enabled precise characterization of drug binding and complex purity.
The results showed clear differences in fluorescence anisotropy between free DOX and DOX-DON complexes, confirming successful drug loading. At higher loading ratios (DOX to DNA base pairs > 0.5), homo-Förster resonance energy transfer (homo-FRET) between closely packed DOX molecules was detected. When the loading ratio exceeded 1, time-resolved anisotropy revealed drug aggregation on the DNA origami structures.
The study also demonstrated that spin-filtration is an efficient and necessary step to remove excess free DOX at loading ratios above 0.1. However, at very high loading levels (>1), some DOX aggregates remained attached to the nanostructures.
Overall, fluorescence anisotropy analysis provided significantly deeper insight compared to conventional fluorescence intensity-based methods. These findings contribute to the rational design of more efficient, controllable, and safer DNA intercalator-based nanocarriers for cancer treatment.
Read the full article here: