Nuclear mechanoadaptation
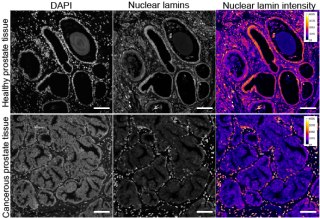
Mechanical forces affect tumorigenesis, invasion and metastatic processes in cancer. Reinforcing, recent findings underline the importance of mechanical forces in nuclear remodelling including nuclear envelope deformation, and changes in nuclear lamina and chromatin organization. In this project, the aim is to study how inter-cellular compressive forces affect nucleus morphology and genetic programming including epithelial-mesenchymal transition and phenotypic reversion in epithelial carcinomas, mainly prostate cancer. The main goal of the study is to understand in detail how nuclear envelope co-regulates chromatin organization under increased compressive force in healthy and prostate cancer cells. Its findings will deepen the current knowledge on the causal connections between compressive force and cell fate. The main tools in this project are in-house built Lego-based cell stretchers, RNAseq, Dam-ID, ATACseq, ATACsee, expansion microscopy and advanced live cell imaging.
Responsible person: AoF postdoctoral fellow Elina Mäntylä, Ph.D.
RPE – retina interaction
Here we investigate the interaction between retinal pigment epithelium (RPE) and retina. RPE resides in the back of the eye and associates closely with the retinal photoreceptor cells. RPE is absolutely crucial for retinal wellbeing as it supports photoreceptor physiology. The interface of the RPE and retina is highly dynamic, e.g. RPE phagocytoses and recycles daily approximately 10% of the photoreceptors. In this project we use different hydrogel and microengineering approaches to investigate this interaction. Project is done in collaboration with Biophysics of the Eye -research group.
Responsible person: Ph.D. student Sanna Korpela, M.Sc. (tech)
Optical control of cellular mechanics
This project aims to combine light responsive materials and optogenetic control of cellular contractility. Here we use light responsive DR1-glass to manipulate the cell-ECM interface topography. Project is done in collaboration with Smart Photonic Materials (SPM) and Biophysics of the Eye -research groups.
Responsible person: Ph.D. student Heidi Peussa, M.Sc. (tech)
