Lue suomeksi täällä.
The study was conducted in collaboration with the NeuroGroup led by Susanna Narkilahti developing neural networks and the Adult Stem Cell Group led by Susanna Miettinen, which develops vascular network. Isosaari works at the NeuroGroup.
Artificial tissues using stem cell technology
Isosaari is delighted with the exceptionally clear results of the study:
“The values typical of blood vessels were all clearly higher when cultured with nerve cells. Even nerve cells did not thrive alone in conditions intended for vasculatures, but together the cell types supported each other’s function. The research is significant, for example, for brain tissue modelling,” she mentions.
The nerve cells used in the study have utilised iPSC stem cell technology, in which cells that are easily isolated from the patient, such as blood cells, have been reprogrammed into stem cells and differentiated into nerve cells with the help of biomolecules. This technology makes it possible to study a number of diseases in the patient. The NeuroGroup examines, for example, the cells of patients with epilepsy and Parkinson’s disease.
Adult adipose tissue and bone marrow stem cells were used to form the vascular network. Samples for stem cell isolation have been obtained from donors at Tampere University Hospital in connection with clinical patient work. In the experiments, stem cells were cultured with blood vessel wall cells obtained from commercial sources.
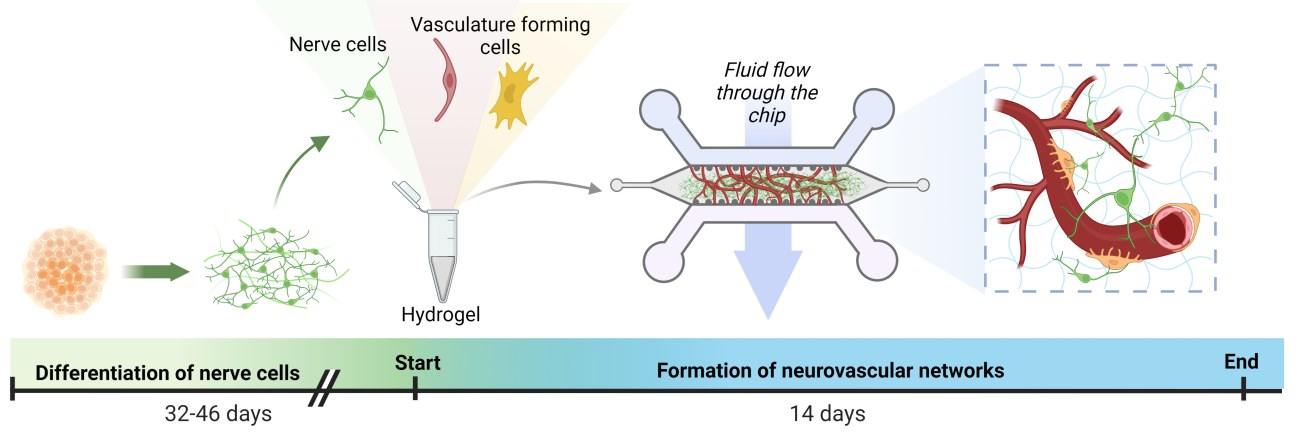
Heart attacks and strokes in cell culture chambers
The Centre of Excellence in Body on-Chip Research consists of six research groups at Tampere University and focuses especially on the study of tissue damage caused by hypoxia in cases such as heart attack or stroke.
The topic has not yet been studied much abroad, which has challenged the Micro- and Nanosystems Group led by Professor Pasi Kallio at the CoEBoC in the development of completely new kinds of tissue chips that regulate oxygen concentration. The group has strong expertise in oxygen concentration regulation. One oxygen-regulating cell chamber has already been commercialised.
Organ-on-chip technology is used to mimic and study human tissues
Organ-on-chip technology may use human cells and create micro-sized environments that mimic the body so that they can be monitored, for example, through imaging. Organ-on-chips can be used to study, for example, the effects of drugs on a tissue.
There is a great need for human-derived tissue models. Nine out of ten new drug candidates are rejected when moving from animal tests to patient trials. The loss of resources is enormous, considering the extensive drug development, often taking ten years, not to mention the ethical challenges of animal testing.
“The functions of the human body can be very different from those of a laboratory animal. A mouse’s heart rate, for example, is 600 beats per minute, compared to human’s 60 beats per minute,” Postdoctoral Research Fellow Hanna Vuorenpää says.
The transformation is now in full swing both commercially and in government agencies. The US Food and Drug Administration (FDA), for example, has stated this year that drugs can be approved for patient trials without animal testing.
Cross-disciplinary collaboration plays a key role
The work requires experts from many fields. Last year, Doctoral Researcher in biotechnology Alma Yrjänäinen, who participated in Isosaari’s research, published a study in which tissue-specific flow-through vasculature was developed for organ-on-chips. Help was needed for this, among others, from microfluidics experts in Kallio’s group.
In addition to organ-on-chip structures and imaging technologies, hydrogels mimicking the tissue environment, for example, are needed around cells. Computer modelling, on the other hand, is used to predict patients’ response to a situation modelled in an organ-on-chip, such as the effect of a drug on heart beating. All these sub-areas are developed in the CoEBoC’s research groups.
In other words, the coexistence of nerves and blood vessels will continue at the CoEBoC for a long time to come.
“Next, we aim to prolong the survivability of vasculature and at the same time complicate the tissue model by adding cells typical of tissues,” Isosaari says.
Isosaari’s and her colleagues’ study was published in the peer-reviewed Cell Communication and Signaling journal is available here.
See also an illustrative video of the study.

