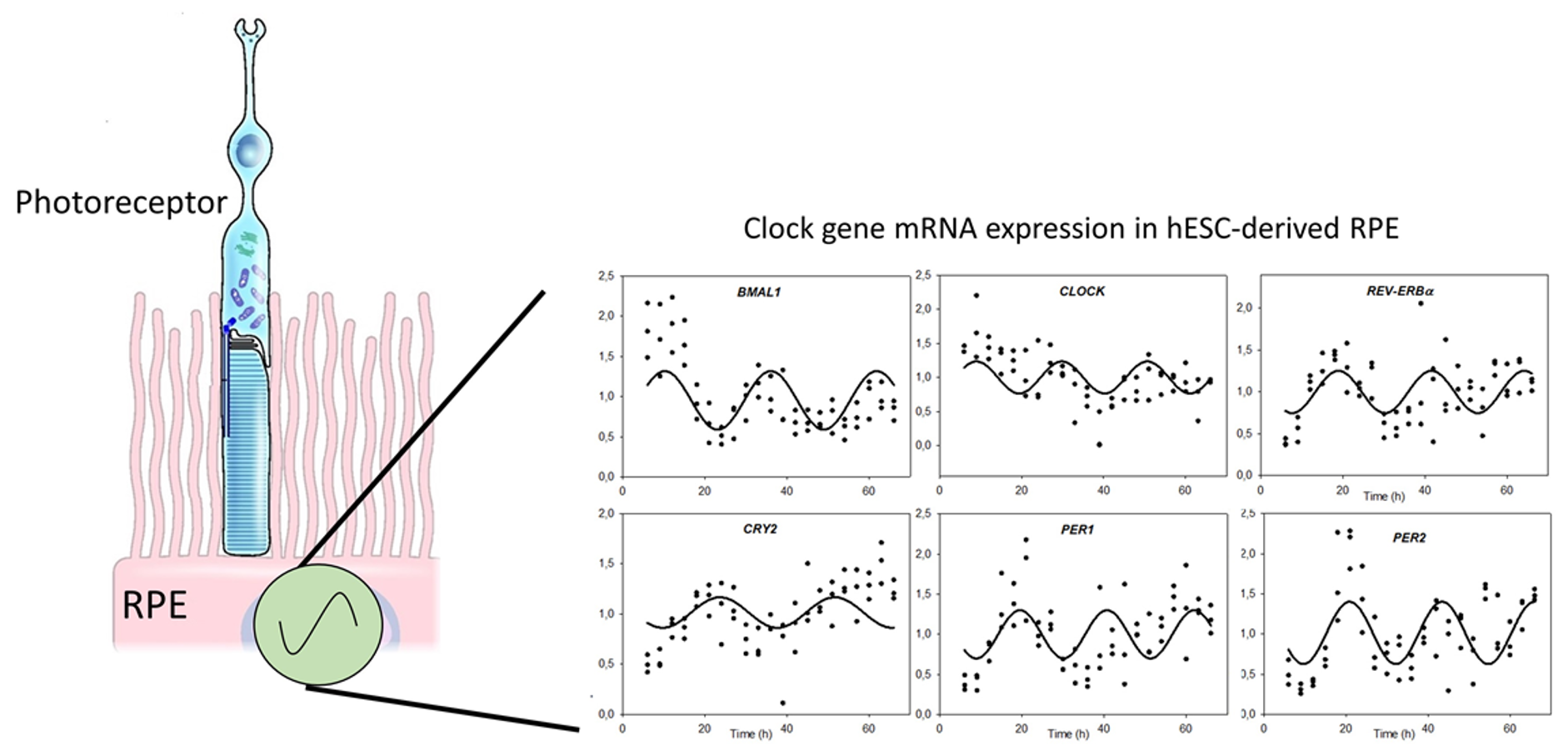
Circadian clocks are endogenous oscillators that control 24-h physiological and behavioral processes. The central clock in mammals located in the suprachiasmatic nuclei (SCN) in the brain integrates environmental light transmitted by the retina, and coordinates the peripheral clocks throughout the body. These oscillations are self-driven on a molecular level via complex interactions of transcriptional and translational feedback loops, with BMAL1 and CLOCK comprising the positive and Period (PER) 1–3 and Cryptochrome (CRY) 1–2 proteins comprising the negative feedback loops. The coordinated activity of the cellular clock gene network drives rhythmic expression of a plethora of clock-controlled genes that, in turn, regulate rhythmic physiology and metabolism.
Circadian clocks regulate a plethora of processes in the RPE including the permeability of the blood-retinal barrier, renewal of photoreceptor outer segments and ion transport. However, little is known about the regulation of the RPE clock. Moreover, most of our knowledge about this clock and its role is inferred from mouse studies. A potential mechanism linking the circadian clock and RPE-function is via the complex interplay of ion channel activities. This possibility is currently under investigation using a variety of techniques including bioinformatics, molecular biology, electrophysiology and confocal microscopy.