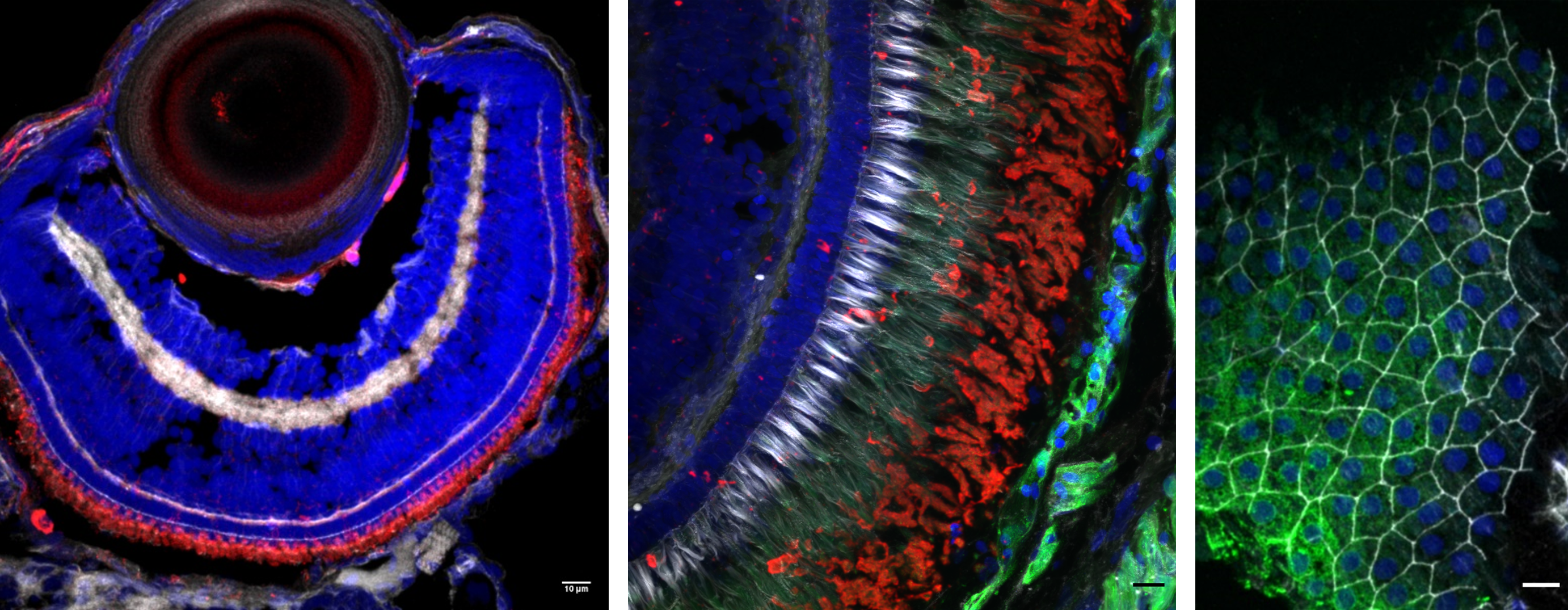
Zebrafish (Danio rerio) is an excellent model to study biological processes and retinal physiology in vivo. Retina-RPE (retinal pigment epithelium) interactions are vital for the correct functioning of the eye and failure in any of these interactive functions can lead to degeneration of the retina and to blinding eye diseases that affect hundreds of millions of people worldwide. Photoreceptor cells shed and renew 10% of their outer segments every day to prevent toxic effects of accumulated photo-oxidative products, requiring efficient phagocytosis controlled by the RPE. Our research aims to resolve individual steps in the critical RPE mediated phagocytosis in live zebrafish to complement the previous ex vivo studies done with rod-dominated animals and in vitro models. We utilize transgenic zebrafish lines together with state-of-the-art imaging techniques to study the crucial retina-RPE interactions in larval and adult zebrafish. In addition, we use histological methods with ex vivo samples.
Recent work in our group has demonstrated how electrical signaling through voltage-gated sodium channels (NaV) is vital for the RPE physiology and regulation of the phagocytosis. However, detailed understanding of how ion channels affect the phagocytosis, including its dynamics, have not yet been resolved. Our aim is to identify different ion channels present in the zebrafish RPE to study their physiological roles and function in the RPE mediated phagocytosis process.