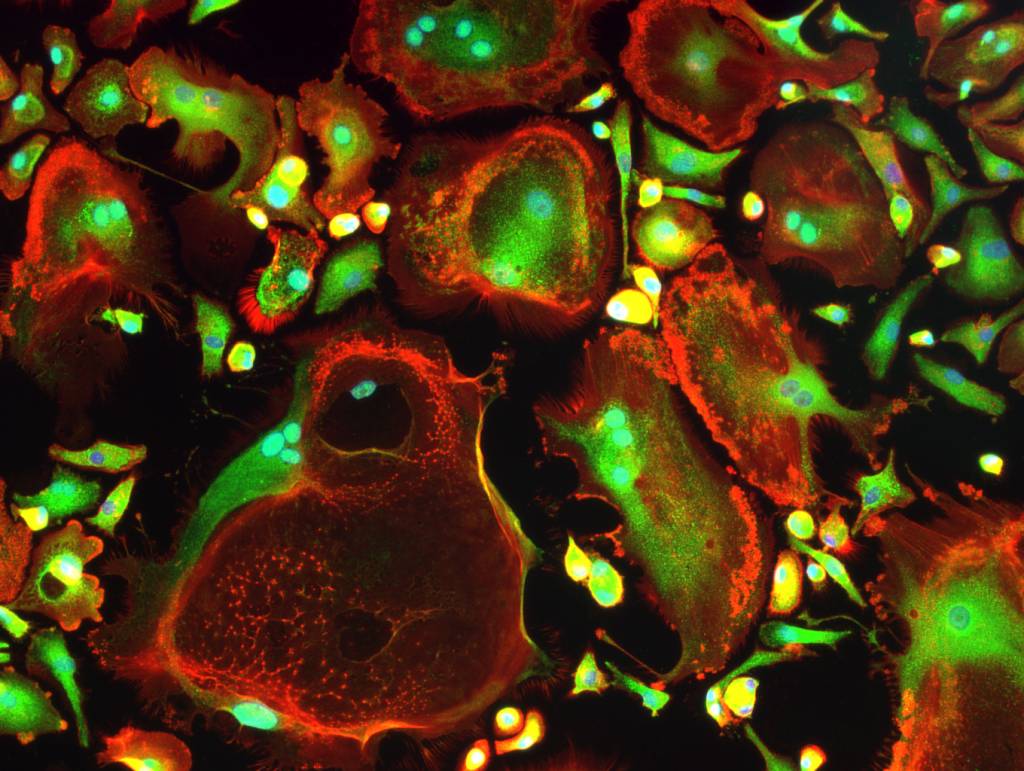
In 2060, ca 155 million Europeans are 65 years of age or older (source: European Commission). Because of the aging population, the incidence of diseases and traumas will increase heavily, posing severe challenges to our health care system. Our group is developing cell and tissue engineering -based solutions to treat medical conditions that currently have very limited treatment options, such as large bone defects. In addition, we develop in vitro models for studying disease mechanisms, novel therapy approaches and drug screening.
Cells and Biomaterials
High quality cells of human origin are the backbone of our group’s research. In the majority of our projects, we use mesenchymal stem/stromal cells from adipose tissue and bone marrow. In addition, we use endothelial cells, epithelial cells, muscle cells, peripheral and cord blood derived mononuclear cells, and many other cell types for our research. Cells are extracted from tissue samples obtained from surgical operations at the Tampere University Hospital. We are grateful for doctors and tissue donors for enabling our research.
Techniques
– Stem and primary cell extraction from tissue samples
– Cell culturing techniques
– Flow cytometry
– Microfluidics
– 3D bioprinting
– Bioimaging
– Immunocytochemistry
– qRT-PCR
– Protein & cytokine arrays
– western blotting
– Seahorse-based analyses for cell respiration
Cells are often combined with biomaterial scaffolds for guiding cell differentiation and to support formation of a 3D microenvironment in tissue engineering applications and in vitro –models.
Major research topics
Adipose stem/stromal cells characteristics and obesity research
Donor characteristics and tissue source might affect cell characteristics and thereby potential for cell-based therapies and other applications. One of our research questions is, how ASC donor obesity affect ASC properties. We have conducted a longitudinal sample collection from obese donors who undergo weight loss treatment in Tampere University Hospital. Using this informative patient material, we characterize the influence of donor obesity and type II diabetes on ASC properties.
Our research combines stem cell and obesity research with immunology. We investigate the capacity of human ASC in regulation of inflammatory responses in co-cultures with human macrophages and T cells. We also characterize the effect of donor obesity on ASC metabolism and cellular respiration capacity. In addition, we characterize the visceral versus subcutaneous adipose tissue of obese donors and investigate how tissue source influences the ASC properties. The final aim of this research is to provide novel therapeutic insights for immunometabolic diseases.
In vitro models
We are developing in vitro tissue models based on human stem and precursor cells. The tissue models are prepared using microfluidic chips, biomaterials, bioreactors and 3D bioprinting. We focus on the development of in vitro –models of vascularization, adipose tissue, bone, bone marrow and skeletal muscle in addition to stroke. With these models, we aim to establish relevant environment for studying disease mechanisms, novel therapy approaches and drug screening.
Our in vitro –model development is tightly connected to the Academy of Finland Centre of Excellence on Body-on-Chip (CoEBoC) Project, which aims to develop a novel multi-tissue model, or “body-on-a-chip” platform. Read more on the project’s website. We are also developing an adult stem cell-based skeletal muscle model for studying metabolic diseases. The project also includes development of electrical stimulation methods, electrically active electrode materials as well as non-invasive characterization methods.
In collaboration with a pharmaceutical company, we are studying the effects of multiple myeloma drugs on BMSC and their interaction with myeloma cells.
Cell differentiation and tissue engineering
Human mesenchymal stem/stromal cells are a promising cell source for regenerative medicine, partly because of their multipotent differentiation capacity. Understanding how cell differentiation is controlled, is vital for development of cell-based therapies. We are studying molecular mechanisms controlling stem cell behavior and differentiation towards adipogenic and osteogenic lineages, especially the role of cell adhesion and actin cytoskeleton in guiding cellular functions.
Biomaterial scaffolds are used to form 3D tissues and to guide cell differentiation. Biomaterials are developed and tested together with our collaborators. First, we study the cytotoxicity of the material, i.e. cell viability and proliferation responses before continuing with application-specific studies such as differentiation responses. We are also studying cell signaling cascades elicited in cells upon attachment on biomaterial surfaces.
Bone tissue engineering
Despite the good healing capacity of bone, large bone defects cannot heal spontaneously. Bone tissue engineering combines biomaterial scaffolds, stem cells and signaling molecules to produce constructs that restore, preserve, or improve the functions of damaged bone tissue. Our group combines adipose or bone marrow derived mesenchymal stem/stromal cells with biomaterial scaffolds for bone tissue engineering. A wide variety of biomaterials such as different compositions of bioactive glasses, ß–tricalcium phosphate, hydroxyapatite, collagen, poly(L-lactide-co-caprolactone) and their composites have been studied by evaluating cellular responses, such as cell attachment, viability, proliferation, and osteogenic differentiation. In addition to biomaterials, we develop other ways of stimulating bone formation such as mechanical loading and the use of growth factors.
Bioprinting
3D printing and bioprinting are important manufacturing techniques for tissue engineering applications. We currently concentrate on developing bioinks and bioprinting techniques for bone and vascular applications. In the project “Let’s make bone” funded by the Olab Thon Foundation, we aim is to utilize 3D printing and bioprinting techniques to reconstruct vascularized bone tissue. The overall goal is to facilitate clinical translation of 3D printing techniques for bone regeneration. The project is performed in collaboration with the University of Bergen.
To strengthen our collaboration with national and international research groups and the industry, we have a new Academy of Finland RDI partnership project for starting a Bioprinting Network. The goal is to tighten the link between national and international industry and research groups, to share knowledge and to provide collaboration opportunities as well as to find solutions for major challenges that limit the clinical translation of 3D bioprinting. The network activities started in the spring of 2021.
Gynecological applications
Our gynecological applications focus on, pelvic organ rolapses (POP), incontinence and regeneration of urothelium and vaginal mucosa.
Pelvic organ prolapse (POP) is a common disorder, which is caused by the weakened support of the pelvic fascia and musculature leading to the prolapse of the pelvic organs. Patient’s own fascia is the primary repair material for POP; however, the recurrence level is high after the primary repair. Similarly to POP, defects of urethra and vaginal mucosa are relatively common and can result from disease, trauma as well as congenital or iatrogenic reasons. They greatly affect patients` quality of life, but their available repair methods are currently highly limited. We are studying the combination of ASC or fibroblasts and urothelial or vaginal epithelial cells to regenerate both epithelial and stromal compartments of urethra and vaginal mucosa. ASC or fibroblasts are studied for POP. In these applications, cells are combined with films, meshes and porous scaffolds made of biomaterials such as poly(L-lactide-co-caprolactone) and poly(trimethylene carbonate) to support tissue regeneration. In addition, biomaterials with and without drug-releasing properties are studied.
Incontinence means an involuntary leakage of urine or feces. Both are devastating conditions that significantly reduce the quality of life. For these conditions, we are developing ASC-based injection therapy, in which the cells are combined with a hydrogel biomaterial.
We are collaboration with Tampere University Hospital to develop solutions for these applications.
Patient treatments
Autologous adipose tissue derived stem/stromal cells have been used in cell and tissue engineering -based therapies to treat large cranio-maxillofacial bone defects as well as urinary incontinence. Currently, we have a National manufacturing license issued by FIMEA for use of autologous adipose stem/stromal cells and biomaterials for large bone defects. Therapies have been carried out in close cooperation with Regea Cell Center and Tissue Bank and clinicians from Tampere University Hospital, Helsinki University Central Hospital, Central Finland Central Hospital and Sahlgrenska Academy.
Main funding sources
Academy of Finland
– Centre of Excellence in Body-on-Chip Research (CoeBoC) https://research.tuni.fi/coeboc/
– 3D Bioprinting Network
Olav Thon Foundation
– Lets’ Make Bone -project
Finnish cultural foundation
The expert responsibility area of the Tampere University Hospital State Research Funding
Tampere University
EDUFI
